 Additional chemical information has also been elucidated from Auger parameters and by using Wagner plots. The use of well characterized standard samples and fitting of the entire peak shape has been shown to increase our ability to accurately identify and (semi) quantify the various species present in mixed oxide/hydroxide systems. A more consistent, practical and effective approach to curve fitting has been developed based on a combination of 1) standard spectra from quality reference samples, 2) a survey of appropriate literature databases and/or a compilation of literature references, 3) specific literature references where fitting procedures are available and 4) theoretical fittings, where available, of multiplet split reference spectra. 
This is particularly true in cases where species show significant multiplet splitting e.g., Cr, Mn, Fe, Co and Ni. 
There are numerous cases in the literature where lack of understanding of these parameters has led to erroneous interpretation of XPS data. Recent work has shown that all the values of the spectral fitting parameters for each specific species, i.e., binding energy (eV), full width at half maximum (FWHM) value (eV) for each pass energy, spin-orbit splitting values and asymmetric peak shape fitting parameters, are not all normally provided in the literature and databases, and are necessary for reproducible, quantitative chemical state analysis. 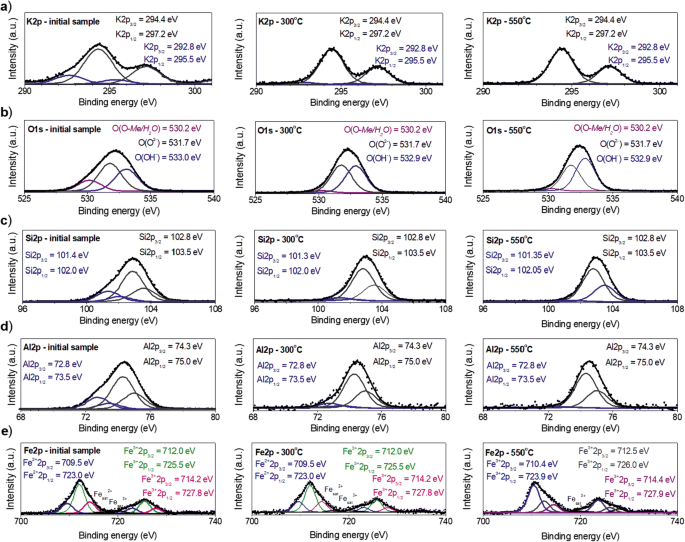
Chemical state X-ray photoelectron spectroscopic (XPS) analysis of first row transition metals and their oxides and hydroxides is challenging due to the complexity of the 2p spectra resulting from peak asymmetries, complex multiplet splitting, shake-up and plasmon loss structure, and uncertain, overlapping binding energies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
